Drug administered to the mother may cross the placenta.
Drug administered to the mother may cross the placenta. Numbers of drug prescribed during pregnancy should be restricted to a minimum with lowest dose for shortest time.
Numbers of drug prescribed during pregnancy should be restricted to a minimum with lowest dose for shortest time. The time period between 35-55 days from LMP is most crucial is may result in missed abortion or has bad effect on the growing baby.
The time period between 35-55 days from LMP is most crucial is may result in missed abortion or has bad effect on the growing baby. USFDA has categorized drugs in categories ABCDX for use in pregnancy.
USFDA has categorized drugs in categories ABCDX for use in pregnancy.
Maternal drug intake and breast feeding
➤ Maternal drug intake may have deleterious effect not only on lactation but also on the baby through the ingested breast milk
➤ Any drug ingested by lactating mother may be present in her breast milk but the amount – concentration are usually low compared to blood
➤ Most drugs are compatible with breast feeding . The physician must therefore consider the benefit of mother and possible danger the baby may face each time drug is prescribed during nursing period.
Maternal medication during pregnancy and fetal hazards
➤ Maternal foetal drug transfer, prior to implantation , ie during embryogenesis drug reach the conceptus through the tubal and uterine section by diffusion effect is usually death in case of survival congenital anomaly exist during organogenesis.
➤ During pregnancy mother and baby represent nonsepratable single unit.
➤ It is important to treat the mother whenever needed while protecting the unborn baby to the greatest possible extent.
➤ During antenatal care what we are give is only nutritional supplements and vaccines.
➤ Women may required treatment for illness during pregnancy for example upper respiratory tract infection, urinary tract infection, malaria, epilepsy, TB, diabetes, blood pressure , preterm labour pain etc.
DRUGS AND CHEMICAL PROVEN TO BE TERATOGENIC
➤ ALCHOHOL : Foetal alcohol syndrome , during pregnancy alleviate fears in mild or occasional drinkers who may terminate pregnancy based on unrealistic perception of risk . Women consuming 2gm/Kg/day during the 1st trimester has 1 to 3 fold high risk for congenital malformation. Alchohol is freely distributed in the milk. She should not breast feed her baby atleast 2 hour after consumption of alchohol
➤ NICOTIN: Do not smoke during lactation it can cause infant colic, decresed milk flow and erlier weaning
➤ STRET DRUGS: It must be avoided such as marijuhana, cocain etc.
Many mothers are required to use drugs during breastfeeding. Almost all drugs transfer into breast milk and this may carry a risk to a breastfed infant. Factors such as the dose received via breast milk, and the pharmacokinetics and effect of the drug in the infant need to be taken into consideration. Problems should not be overstated however, as many drugs are considered ‘safe’ during breastfeeding.
Transfer of drugs into breast milk is influenced by protein binding, lipid solubility and ionisation
Nearly all drugs transfer into breast milk to some extent. Notable exceptions are heparin and insulin which are too large to cross biological membranes. The infant almost invariably receives no benefit from this form of exposure and is considered to be an ‘innocent bystander’. Drug transfer from maternal plasma to milk is, with rare exceptions, by passive diffusion across biological membranes. Transfer is greatest in the presence of low maternal plasma protein binding and high lipid solubility. In addition, milk is slightly more acidic than plasma (pH of milk is approximately 7.2 and plasma is 7.4) allowing weakly basic drugs to transfer more readily into breast milk and become trapped secondary to ionisation. Milk composition varies within and between feeds and this may also affect transfer of drugs into breast milk. For example, milk at the end of a feed (hindmilk) contains considerably more fat than foremilk and may concentrate fat-soluble drugs.
Transfer of drugs into breast milk is most commonly described quantitatively using the milk to plasma (M/P) concentration ratio. The accuracy of this value is improved if it is based on the area under the concentration-time curves (AUC) of the drug in maternal milk and plasma (M/PAUC).
Calculation of infant exposure to drugs can be used to help guide safe use
The infant’s dose (Dinfant) received via milk can be calculated using the maternal plasma concentration (Cmaternal), M/PAUC ratio and the volume of milk ingested by the infant (Vinfant):
Dinfant (mg/kg/day) = Cmaternal (mg/L) x M/PAUC x Vinfant (L/kg/day)
The volume of milk ingested by infants is commonly estimated as 0.15L/kg/day. The infant dose (mg/kg) can then be expressed as a percentage of the maternal dose (mg/kg). An arbitrary cut-off of 10% has been selected as a guide to the safe use of drugs during lactation. Drugs such as lithium (infant dose as high as 80% of the weight-adjusted maternal dose) and amiodarone (infant dose up to 50%) should be avoided due to high infant exposure and potential for significant toxicity. For drugs with greater inherent toxicity such as cytotoxic agents, ergotamine, gold salts, immunosuppressives and isotretinoin, the cut-off of 10% is too high and breastfeeding is contraindicated.
As a general rule, maternal use of topical preparations such as creams, nasal sprays or inhalers would be expected to carry less risk to a breastfed infant than systemically administered drugs. This is due to lower maternal concentrations and therefore lower transfer into breast milk. However, the risk to the infant must be considered in relation to the toxicity of the drug used, the dosage regimen and the area of application. For example, use of corticosteroids nasal sprays or inhalers in standard doses would be considered compatible with breastfeeding.
Other factors to consider in conjunction with the infant’s dose include the pharmacokinetics of the drug in the infant. Generally, drugs that are poorly absorbed or have high first-pass metabolism are less likely to be problematical during breastfeeding. For example, gentamicin is highly hydrophilic and is very poorly absorbed when administered orally. Should any gentamicin be ingested via breast milk, it is unlikely to be absorbed.
Drug clearance in the infant is a particularly important consideration and premature infants have a severely limited ability to clear drugs. Within a few days of delivery, term infants have glomerular filtration rates approximately one-third of adult values after adjusting for difference in body surface area, and premature infants have even more impaired clearance (see Table 1). Generally, adult glomerular filtration rates (adjusted for the difference in surface area) are attained by five to six months of age. Metabolic processes such as phase 1 oxidation and phase 2 glucuronidation are also impaired in the neonate. Drugs subject to high first-pass metabolism may have higher oral availability in premature or term infants due to impaired ability to metabolise on first-pass. Adult metabolic capacity is attained towards the latter part of the infant’s first year of life. The following table is useful for estimating infant clearance.
Table 1: Approximate clearance values at different ages
| Post-conceptual age | Clearance of drug (compared with adults) |
|---|---|
| 24-28 weeks | 5% |
| 28-34 weeks | 10% |
| 34-40 weeks | 33% |
| 40-44 weeks | 50% |
| 44-68 weeks | 66% |
| > 68 weeks | 100% |
The overall risk of a drug to a breastfed infant depends on the concentration in the infant’s blood and the effects of the drug in the infant. If, after assessment of the risks and benefits, the decision is made to breastfeed while the mother is using a drug, the infant should be monitored for adverse effects such as failure to thrive, irritability and sedation. However, it is difficult to identify adverse reactions occurring in neonates. Feeding immediately prior to a dose may help to minimise infant exposure as concentrations in milk are likely to be lowest towards the end of a dosing interval. However, for some drugs, milk concentrations lag behind plasma concentrations.
For drugs that have an infant dose greater than the arbitrary cut-off of 10% of the weight-adjusted maternal dose, it may be reasonable to reduce infant exposure by alternating breast and bottle-feeding. For drugs that are not considered safe in breastfeeding, breast milk may be expressed and discarded for the treatment duration. Breastfeeding may be resumed after the drug has been eliminated from the maternal blood stream. A period of approximately four half-lives will reduce maternal concentrations to around 10% of steady-state concentrations.
Safety assessment of some frequently used drugs
A discussion of the safety of the more commonly used drugs is provided below. The data must be assessed in conjunction with information on the maternal dose and therefore probable maternal concentrations, the age of the infant and their likely ability to eliminate the drug. In general, if the infant dose as a percentage of the maternal dose (corrected for weight) is close to 1%, the drug can be considered ‘safe’ regardless of infant age. For drugs where the weight-adjusted dose is closer to 10%, the infant clearance should also be taken into account (see Table 1). For example, if the weight-adjusted infant dose is 10% but the infant is premature, the lower clearance will mean that the infant concentrations may be well above those expected.
Analgesics:
Analgesics such as paracetamol, ibuprofen, naproxen and codeine are considered to be ‘safe’, due to low transfer into breast milk and few problems with extensive usage. Transfer of aspirin into breast milk appears to be low but it is best avoided due to the theoretical risk of Reye’s syndrome. Sumatriptan has a short half-life of approximately two hours and infant exposure can be almost completely avoided by expressing and discarding breast milk for approximately eight hours after dosing. Limited data on tramadol suggest low transfer into breast milk although where possible, it would be preferable to use agents which are more established such as codeine and paracetamol. Morphine is usually considered ‘safe’ because of low transfer into milk, and high first-pass metabolism.
Anthelminthics:
There does not appear to be any data on the transfer of mebendazole or pyrantel embonate into human breast milk although these agents are generally considered to be ‘safe’ due to poor absorption from the gastrointestinal tract.
Antibiotics:
Antibiotics such as penicillins, cephalosporins and macrolides are considered to be compatible with breastfeeding although there are theoretical risks of alterations to infant bowel flora and allergic sensitisation.
The safety of metronidazole is controversial due to the possibility of high transfer into breast milk. The weight-adjusted infant dose may be as high as 36% of the maternal dose indicating that infant exposure may be higher than the arbitrary cut-off of 10%. Techniques that may be considered for minimising infant exposure include choosing an alternative antibiotic such as amoxycillin/clavulanic acid (if appropriate), alternating breast and bottle feeding, or withholding breastfeeding during the treatment course. If breastfeeding is to be withheld, the mother should be encouraged to continue to express breast milk while on the antibiotic course but to discard the milk. This will help to maintain lactation and enable the mother to resume breastfeeding at the end of the course.
The transfer of tetracyclines into breast milk is low but they are usually avoided due to the possible risks of inhibiting bone growth or causing dental staining. Fluoroquinolones should also be avoided in breastfeeding as they have been reported to cause arthropathies in immature animals. Sulphonamides such as sulphamethoxazole are unlikely to be problematical in most situations but are best avoided in infants with hyperbilirubinaemia or glucose-6-phosphate dehydrogenase deficiency.
Anticoagulants:
Heparins (unfractionated and low molecular weight) are considered ‘safe’ since these agents have a large molecular weight and do not cross into breast milk to a significant extent. They are also poorly absorbed. Warfarin is also considered to be compatible with breastfeeding as transfer is low, and adverse effects and changes in prothrombin time have not been detected in breastfed infants. However, it would be prudent to monitor the infant’s prothrombin time during treatment.
Anticonvulsants:
Carbamazepine, phenytoin and sodium valproate are generally considered to be compatible with breastfeeding although the infant should be observed for evidence of central nervous system depression. Available data on the safety of lamotrigine in breastfeeding suggest that transfer into breast milk may be considerable and therapeutic concentrations have been detected in breastfed infants. There are insufficient published data to comment on the safety of gabapentin in breastfeeding.
Antidepressants:
Selective serotonin reuptake inhibitors (SSRIs) transfer into breast milk to varying extents. Paroxetine is reported to have the lowest transfer into breast milk (weight-adjusted infant dose 1-3%). Fluoxetine transfers to a greater extent (weight-adjusted infant dose ≤ 14%) and its active metabolite, norfluoxetine, has a long half-life of one to two weeks and may accumulate in a breastfed infant. Data on citalopram (weight-adjusted infant dose approximately 5%) suggest that the relative infant dose of citalopram is intermediate between paroxetine and fluoxetine. Based on these data, paroxetine is the preferred SSRI in breastfeeding women.
Most tricyclic antidepressants are considered to be compatible with breastfeeding due to low transfer into breast milk and this is supported by extensive usage data. Moclobemide has low-transfer into breast milk and is considered compatible with breastfeeding.
Antihistamines:
Agents such as promethazine, dexchlorpheniramine and diphenhydramine are considered to be safe through extensive usage, although it would be prudent to monitor for evidence of sedation or irritability in the infant. There is less data on the non-sedating antihistamines, although loratadine and fexofenadine are likely to be safe due to low transfer into milk.
Benzodiazepines:
Sporadic use of benzodiazepines with a short plasma half-life such as midazolam and temazepam is unlikely to be problematical due to low quantities transferred into breast milk. Agents with a long half-life such as diazepam may accumulate in the infant with prolonged exposure and may be associated with lethargy, poor suckling and reduced weight gain.
Decongestants:
A short course of pseudoephedrine (weight-adjusted dose < 4%) is unlikely to be problematical. However, topical decongestant nasal sprays or drops are usually preferred due to lower infant exposure.
Social drugs:
These have particular problems because the dose and pattern of usage are uncontrolled. In addition most have relatively high infant doses. Infant exposure following maternal ethanol ingestion may be as high as 20% and has been associated with impaired psychomotor development. Alcohol consumption should be minimised during lactation (e.g. by withholding breastfeeding for about two hours after ingestion of a standard alcoholic drink).
Caffeine exposure may be as high as 34% of the weight-adjusted maternal dose and side effects such as restlessness and irritability have been reported in infants exposed via breast milk.
Nicotine has been detected in the plasma of breastfed infants, and smoking is best avoided by breastfeeding mothers. The use of nicotine replacement therapy (e.g. transdermal delivery systems) in breastfeeding mothers should be considered in terms of risks and benefits. However, as a general rule, the short-term use of nicotine replacement therapy is far preferable than continued smoking.
Drugs affecting milk:
Drugs can affect milk secretion or composition by affecting factors such as mammary gland development, milk secretion and hormonal regulation of lactation. Prolactin is necessary for human milk secretion and may be affected by drug use. Dopamine agonists such as cabergoline reduce prolactin and are sometimes used therapeutically to stop lactation. Dopamine antagonists such as metoclopramide and most antipsychotics may increase prolactin (see article on Hyperprolactinaemia With Antipsychotics) and milk production. Other drugs that have been associated with causing hyperprolactinaemia include SSRIs and opioids.
ID = insufficient data
| Drug | M/PAUC | % maternal dose | Comments | |
|---|---|---|---|---|
| Acid-suppressants: | ||||
| Cimetidine | 1.7-5.8 | 5.4-6.7 | Avoid in favour of safer alternatives with lower potential for side effects. May accumulate in milk due to active transport. |
|
| Famotidine | 1.5 | 1.6 | Probably safe. | |
| Ranitidine | 2.8 | 5.0-7.8 | Probably safe when restricted to sporadic doses or a single dose at night-time. May accumulate in milk due to active transport. |
|
| Analgesics: | ||||
| Aspirin | 0.06 | 3.2 | Avoid due to possible association with Reye’s syndrome. | |
| Codeine | 2.16 | 6.8 | Considered safe. | |
| Ibuprofen | 0 | < 0.6 | Considered safe. Not detected in milk. | |
| Indomethacin | 0.37 | < 1.0 | Considered safe. One case of seizures (causality questionable). | |
| Mefenamic acid | ID | 0.3 | Probably safe. | |
| Methadone | 0.47 | 2.2 | Considered safe in methadone maintenance as 60% of infants born to mothers in maintenance programmes develop symptoms of withdrawal. |
|
| Morphine | 2.46 | 0.4 | Considered safe. | |
| Naproxen | ID | 1.1 | Probably safe. | |
| Nefopam | ID | 0.4 | Probably safe. | |
| Piroxicam | ID | 5-10 | Use a NSAID with a shorter half-life where possible. | |
| Paracetamol | 0.8 | 2.9-7.9 | Considered safe. | |
| Sumatriptan | 4.1-5.7 | 0.3-6.7 | Exposure limited by low oral availability in term infants. Expressing for 8 hours post-dose will almost completely avoid exposure. |
|
| Antibiotics: | ||||
| Aminoglycosides | ||||
| Gentamicin | 0.17 | 2.2 | Considered compatible with breastfeeding due to low transfer and low oral availability. |
|
| Cephalosporins |
|
Considered safe. Low transfer into milk. Third generation cephalosporins have greater potential to alter bowel flora. |
||
| Cefaclor | ID | 0.7 | ||
| Cefalexin | 0.09 | 0.5-1.2 | ||
| Cefotaxime | ID | 0.3 | ||
| Ceftriaxone | 0.04 | 0.7-4.7 | ||
| Fluoroquinolones | ||||
| Ciprofloxacin | 2.17 | 4.8 | Avoid fluoroquinolones due to theoretical risk of arthropathies. | |
| Macrolides |
|
Considered safe. May alter bowel flora. | ||
| Clarithromycin | 0.25 | 1.8 | ||
| Erythromycin | 0.41 | 2.1 | ||
| Penicillins |
|
Considered safe. Note: although amoxycillin/clavulanic acid combination is used extensively in lactation, there are no published data on the safety of clavulanic acid. |
||
| Amoxycillin | ID | 0.7 | ||
| Benzylpenicillin | 0.37 | 0.8 | ||
| Phenoxymethyl-penicillin | ID | 0.25 | ||
| Tetracyclines |
|
Avoid tetracyclines where feasible due to the possible risks of dental staining and adverse effects on bone development. |
||
| Minocycline | ID | 3.6 | ||
| Tetracycline | 0.58 | 4.8 | ||
| Others | ||||
| Aciclovir | ID | 1.1-1.2 | Considered safe. No adverse effects noted in breastfed infants. | |
| Fluconazole | 0.75 | 11 | Potential for accumulation particularly in premature infants. | |
| Metronidazole | 0.9-1.1 | 0.1-36.0 | Controversial as exposure may be high. With high doses consider expressing and discarding milk. |
|
| Nitrofurantoin | ID | 0.6-6.0 | Avoid in G6PD-deficient infants (due to the risk of haemolysis). | |
| Sulphamethoxazole & Trimethoprim (i.e. co-trimoxazole) |
0.1 1.26 |
2-2.5 3.8-5.5 |
Avoid suphaemethoxazole in infants with hyperbilirubinaemia and G6PD deficiency. | |
| Anticoagulants | ||||
| Warfarin | 0 | < 4.4 | Probably safe. No changes in prothrombin times detected in breastfeeding infants. Monitor prothrombin time. |
|
| Anticonvulsants: | ||||
| Carbamazepine | 0.36-0.39 | 2.8-7.3 | Considered safe. Monitor for sedation, poor suckling. | |
| Lamotrigine | ID | 10-22 | Concentrations in breastfed infants have been consistent with those expected to produce clinical effect. Best to avoid. |
|
| Phenobarbitone | ID | 23-156 | Avoid due to high infant exposure. | |
| Pheyntoin | 0.13-0.18 | 3.0-7.2 | Considered safe. Observe for sedation, poor suckling. One report of methaemoglobinaemia, poor suckling and sedation. |
|
| Sodium valproate | 0.05 | 1.8 | Considered safe at low doses. High doses may increase the risk of hepatitis. | |
| Vigabatrin | ID | <1% | Avoid until further data are available. | |
| Antidepressants: | ||||
| Tricyclics: |
|
Probably safe. Negligible or no concentrations detected in breastfed infants. | ||
| Amitriptyline | 0.83 | 0.6-0.9 | ||
| Desipramine | ID | 0.5-1.0 | ||
| Dothiepin | 0.8-1.6 | 0.2-1.5 | ||
| Doxepin | ID | 0.01 | ||
| Imipramine | ID | 0.13 | ||
| Nortriptyline | ID | 0.53 | ||
| SSRIs | ||||
| See text | ||||
| Others | ||||
| Moclobemide | 0.72 | 1.6 | Probably safe. | |
| Antiemetics: | ||||
| Domperidone | ID | 0.05 | Probably safe. May increase milk secretion. | |
| Metoclopramide | ID | 4.7-11.3 | Low dose or sporadic use probably safe. May increase milk secretion. | |
| Antihistamines: | ||||
| Loratadine | 1.2 | 0.7 | Probably safe. No adverse effects reported in infants. | |
| Triprolidine | 0.53 | 0.9 | Considered safe. | |
| Antipsychotics: |
|
Probably safe. May increase milk secretion. Monitor infant for sedation, irritability etc. |
||
| Chlorpromazine | ID | 0.2 | ||
| Flupenthixol | ID | 0.5-0.8 | ||
| Haloperidol | ID | 0.15-2.0 | ||
| Cardiovascular: | ||||
| Amiodarone | ID | 37 | Avoid in breastfeeding. | |
| Atenolol | 2.3-4.5 | 5.7-19.2 | Avoid in favour of antihypertensives with lower infant exposure. | |
| Captopril | 0.03 | 0.014 | Considered safe. | |
| Digoxin | 0.6-0.9 | 2.3-5.6 | Considered safe. | |
| Diltiazem | 0.98 | 0.9 | Unlikely to be problematical in breastfeeding. | |
| Enalapril | 0.02 | < 0.1 | Considered safe. | |
| Metoprolol | 2.8-3.6 | 1.7-3.3 | Probably safe. | |
| Nadolol | 4.6 | 5.1 | Consider choosing a beta-blocker with a lower infant dose, if feasible. | |
| Propranolol | 0.32-0.76 | 0.2-0.9 | Probably safe. | |
| Quinapril | 0.12 | 1.6 | Considered safe. | |
| Verapamil | 0.6 | 0.14-0.84 | Considered safe. | |
| Sedatives/hypnotics: | ||||
| Clonazepam | ID | 1.5-3.0 | Short-term use of low doses is probably safe. | |
| Diazepam | 0.16 | 2.0-2.3 | Reasonable to breastfeed after a low single dose but potential for accumulation with prolonged use. Sedation has been reported in breastfed infants. |
|
| Lorazepam | ID | 2.2 | Short-term use of low doses is probably safe. | |
| Midazolam | 0.16 | 0.7 | Short-term use of low doses is probably safe. | |
| Nitrazepam | ID | ID | Short-term use of low doses is probably safe. Potential for accumulation with prolonged administration. |
|
| Zopiclone | 0.5 | 4.1 | Short-term use of low doses is probably safe. | |
| Social Drugs: | ||||
| Cannabis (THC) | ID | ID | Avoid as long-term effects are unknown. | |
| Caffeine | 0.5-0.8 | 0.6-21.0 | Low intake probably safe. Restlessness and irritability documented. Prolonged half-life (80-100 hours) in neonates. |
|
| Ethanol | 0.9 | 3-4 | Occasional low usage probably safe. Chronic intake may be associated with impairment of psychomotor development. Consider withholding breastfeeding for 1-2 hours per standard drink. |
|
| Nicotine | 2.92 | ID | Cigarette smoking should be avoided due to health hazards associated with smoking. Use of nicotine patches may be considered compatible with breastfeeding and is favoured over smoking. |
|
| Miscellaneous: | ||||
| Ethinyloestradiol | ID | 0.3 | May suppress lactation. | |
| Levonorgestrel | ID | 1.1 | Considered safe. | |
| Medroxyprogesterone | ID-0.72 | 3.4-5.0 | Considered safe. | |
| Norethisterone | ID-0.26 | 0.02-1.9 | Considered safe. | |
| Prednisone | ID | 0.26 | Short courses of low doses (≤ 20mg daily) are probably safe. Note: there are insufficient data on other systemic corticosteroids (e.g. betamethasone, dexamethasone). |
|
| Pseudoephedrine | 2.5 | 4.0 | Low doses or sporadic use probably safe. | |
| Sulphasalazine | ID | 1.2-7.0 | Avoid in infants with hyperbilirubinaemia or G6PD deficiency. | |
Safe Medications during Breastfeeding
Please Note: Always consult your doctor or healthcare specialist for medical advice in relation to drug use while pregnant or breastfeeding.
Many mothers need to take medications during breastfeeding. Although many drugs are safe to use when you’re breastfeeding, most will get into your milk to some degree and may even affect your milk supply. To be safe, check with your child’s doctor before taking any kind of medication, even over-the-counter drugs. If you have more questions about how a drug you’re taking might affect your breast milk or your baby, check our pregnancy warnings and breastfeeding warnings pages.
Special precautions may be needed in preterm (premature) infants.
If possible, take medications that are given only once a day right after a feeding when your baby will have the longest period without nursing; for many women this the last feeding of the night before the infants bedtime.
Some of pregnant women take prescription or nonprescription (over-the-counter) drugs or use social drugs (such as tobacco and alcohol) or illicit drugs at some time during pregnancy, and use of drugs during pregnancy is increasing. In general, drugs should not be used during pregnancy unless absolutely necessary because many can harm the fetus. About 2 to 3% of all birth defects result from drugs that are taken to treat a disorder or symptom.
Sometimes drugs are essential for the health of the pregnant woman and the fetus. In such cases, a woman should talk with her doctor or other health care practitioner about the risks and benefits of taking the drug. Before taking any drug (including over-the-counter drugs) or dietary supplement (including medicinal herbs), a pregnant woman should consult her health care practitioner. A health care practitioner may recommend that a woman take certain vitamins and minerals during pregnancy.
Drugs taken by a pregnant woman reach the fetus primarily by crossing the placenta, the same route taken by oxygen and nutrients, which are needed for the fetus’s growth and development. Drugs that a pregnant woman takes during pregnancy can affect the fetus in several ways:
• They can act directly on the fetus, causing damage, abnormal development (leading to birth defects), or death.
• They can alter the function of the placenta, usually by causing blood vessels to narrow (constrict) and thus reducing the supply of oxygen and nutrients to the fetus from the mother. Sometimes the result is a baby that is underweight and underdeveloped.
• They can cause the muscles of the uterus to contract forcefully, indirectly injuring the fetus by reducing its blood supply or triggering preterm labor and delivery.
• They can also affect the fetus indirectly. For example, drugs that lower the mother’s blood pressure may reduce blood flow to the placenta and thus reduce the supply of oxygen and nutrients to the fetus.
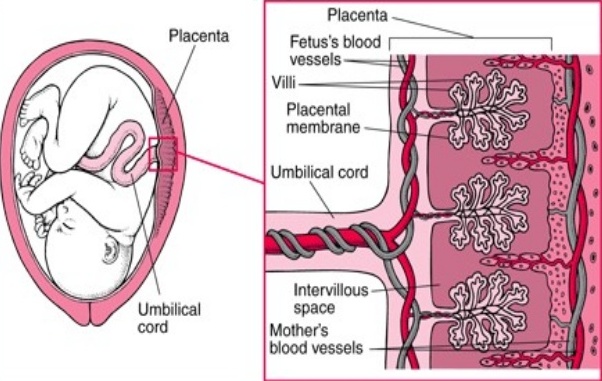
How Drugs Cross the Placenta
Some of the fetus’s blood vessels are contained in tiny hairlike projections (villi) of the placenta that extend into the wall of the uterus. The mother’s blood passes through the space surrounding the villi (intervillous space). Only a thin membrane (placental membrane) separates the mother’s blood in the intervillous space from the fetus’s blood in the villi. Drugs in the mother’s blood can cross this membrane into blood vessels in the villi and pass through the umbilical cord to the fetus.
How a drug affects a fetus depends on the fetus’s stage of development and the strength and dose of the drug.